 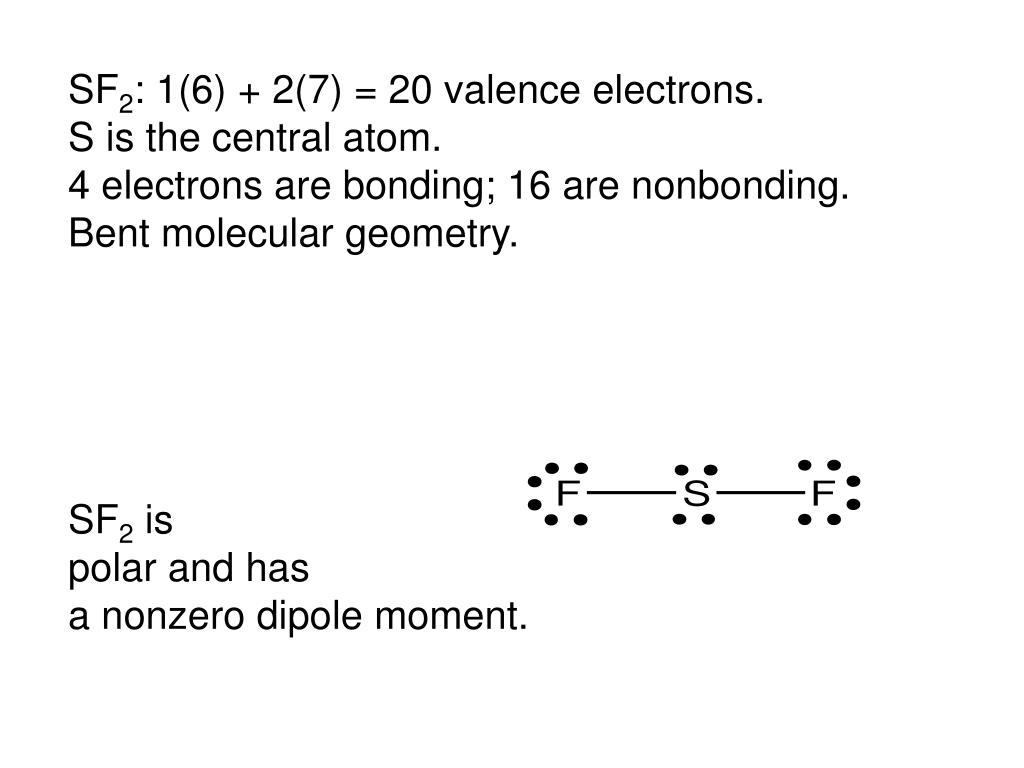
The molecular shape of the C2Cl2 molecule is linear. The two lone pairs of electrons push the Fluorine atoms downwards due to the repulsive forces, and as a result, the shape of this molecule is bent. In the Lewis Structure of SF2, the central atom forms two bonds with two Fluorine atoms and has two lone pairs of electrons.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
